AARP Eye Center
Meningitis Outbreak From Shots For Back Pain
By Candy Sagon, October 5, 2012 08:49 AM
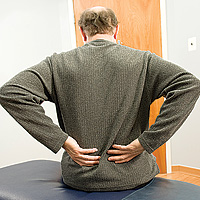
A popular treatment for severe back pain - steroid shots in the lower back because of ruptured disks or spinal stenosis - has been linked to a growing outbreak of a rare and deadly fungal meningitis from contaminated injections.
As the number of cases and deaths continued to climb this week, lawmakers called for a Congressional inquiry into pharmacies of the kind that made the medicine and new laws to ensure tighter federal oversight of their operations, the New York Times reported.
Health providers are scrambling to notify patients in nearly two dozen states that the routine steroid injections they received for back pain between May and September may have been contaminated with a fungus causing a potentially fatal brain infection.
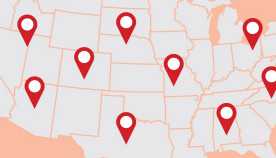
You can check to see if your health facility is on the list of those receiving the contaminated medication by clicking on this page from the Centers for Disease Control and Prevention (CDC).
So far, 170 people in 11 states - Tennessee, Virginia, Maryland, Florida, North Carolina, Michigan, Ohio, Minnesota, Indiana, Idaho and New Jersey - have contracted fungal meningitis, and 14 of them have died, according to the Centers for Disease Control and Prevention. One of the latest victims was a 70-year-old man from Florida, Reuters reported.
The CDC is urging doctors to contact all patients who received potentially contaminated injections starting May 21, USA Today reported. Symptoms can take anywhere from a few days to a month to develop.
"All patients who may have received these medications need to be tracked down immediately," said CDC medical officer Benjamin Parks. "It is possible that if patients with infection are identified soon and put on appropriate anti-fungal therapy, lives may be saved."
Meningitis is an inflammation of the lining of the brain and spinal cord. Symptoms include a severe headache, nausea, dizziness, slurred speech and fever. Fungal meningitis is not contagious like the more common forms, bacterial and viral meningitis. It is caused by a fungus often found in leaf mold.
In an analysis of the health crisis, the Times reported that this was "a calamity waiting to happen - the result of a lightly regulated type of drug production that had a troubled past colliding with a popular treatment used by millions of Americans a year."
The steroid solution was not made by a major drug company, the newspaper said, but was custom-made by a pharmacy in Framingham, Mass., called the New England Compounding Center. Compounding pharmacies make their own drug products, which are not approved by the Food and Drug Administration.
(Updated Thurs., Oct. 11, 2012)
Photo: National Institutes of Health